

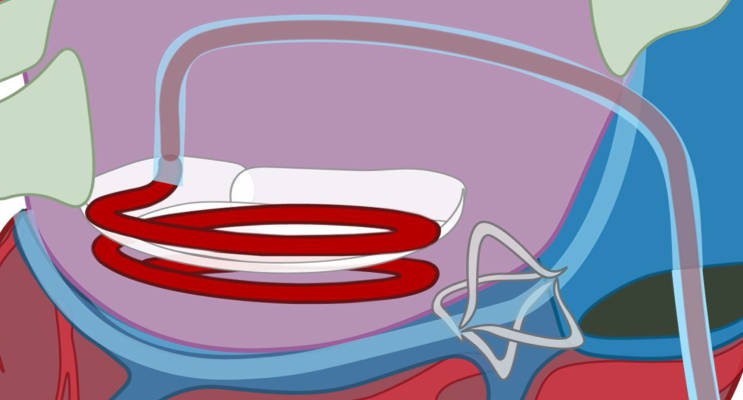


HVR Cardio’s patented helix ring technology can be applied to interventional cardiology as well as to cardiac surgery.
HVR Cardio’s innovative helix ring technology is designed to mimic mitral valve geometry. The helix ring is rotated from posterior commissura (the area where the leaflets come together in the mitral annulus) into its intended position. The rotation ensures that the implant is correctly positioned, having one of the helix loops above and the other one below the mitral annulus. This results in faster, easier and more reliable placement of the ring and restoration of the geometry of the mitral annulus.
